Solanesol from tobacco
solanesol
CAS: 13190-97-1
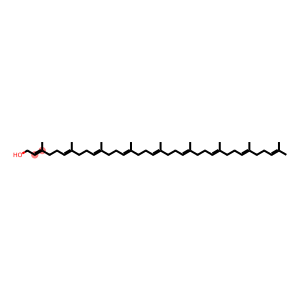
Molecular Formula: C45H74O
Solanesol from tobacco - Names and Identifiers
| Name | solanesol |
| Synonyms | Solanasol solanesol Farnesylfarnesylfarnesol Solanesol from tobacco 3,7,11,15,19,23,27,31,35-nonamethylhexatriaconta-2,6,10,14,18,22,26,30,34-nonaen-1-ol (2E,6E,10E,14E,18E,22E,26E,30E)-3,7,11,15,19,23,27,31,35-nonamethylhexatriaconta-2,6,10,14,18,22,26,30,34-nonaen-1-ol (2E,6E,10E,14E,18E,22E,26E,30E)-3,7,11,15,19,23,27,31,35-Nonamethyl-2,6,10,14,18,22,26,30,34-hexatriacontanonaene-1-ol (2E,6E,10E,14E,18E,22E,26E,30E,)-3,7,11,15,19,23,27,31,35-Nonamethyl-2,6,10,14,18,22,26,30,34-hexatriacontanonaen-1-ol 2,6,10,14,18,22,26,30,34-Hexatriacontanonaen-1-ol, 3,7,11,15,19,23,27,31,35-nonamethyl-, (2E,6E,10E,14E,18E,22E,26E,30E)- |
| CAS | 13190-97-1 |
| EINECS | 603-532-7 |
| InChI | InChI=1/C45H74O/c1-37(2)19-11-20-38(3)21-12-22-39(4)23-13-24-40(5)25-14-26-41(6)27-15-28-42(7)29-16-30-43(8)31-17-32-44(9)33-18-34-45(10)35-36-46/h19,21,23,25,27,29,31,33,35,46H,11-18,20,22,24,26,28,30,32,34,36H2,1-10H3/b38-21+,39-23+,40-25+,41-27+,42-29+,43-31+,44-33+,45-35+ |
| InChIKey | AFPLNGZPBSKHHQ-MEGGAXOGSA-N |
Solanesol from tobacco - Physico-chemical Properties
| Molecular Formula | C45H74O |
| Molar Mass | 631.07 |
| Density | 0.889±0.06 g/cm3(Predicted) |
| Melting Point | 33°C |
| Boling Point | 685.6±24.0 °C(Predicted) |
| Flash Point | 130.2°C |
| Solubility | Insoluble in water, slightly soluble in methanol, ethanol, soluble in acetone, chloroform, hexane |
| Vapor Presure | 9.21E-22mmHg at 25°C |
| Appearance | Pale yellow powder |
| Color | Off-White to Brown Waxy to |
| Merck | 14,8703 |
| pKa | 14.42±0.10(Predicted) |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Stability | Stable, but may be heat sensitive - store cold. Combustible. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.506 |
| MDL | MFCD00070279 |
| Physical and Chemical Properties | Melting Point 33°C |
| Use | Mainly used for the synthesis of pharmaceutical Coenzyme Q10 and vitamin K2 |
Solanesol from tobacco - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| RTECS | MP5366666 |
| HS Code | 29052900 |
Solanesol from tobacco - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| market research | solanesol is a natural product widely found in higher plants, mammals and microorganisms. it is a waxy white solid with a melting point of 33~42.5 ℃, insoluble in water and soluble in various organic solvents. The natural product is an all-trans isomer. In 1956, it was first isolated from plants by R.L.Rowland et al.. In I959, Frickson et al. used nuclear magnetic resonance spectroscopy to prove that solanesol is composed of 9 isoprene units, and it was later discovered that the content of solanesol in tobacco leaves is relatively high, about 0.3-0.5%. Some people have also studied that the content of solanesol in Yunnan tobacco leaves is between 0.5-0.8%. Now waste tobacco leaves are generally used as raw materials, first, the crude solanesol (including 17% solanesol) is extracted, and the fine solanesol (content 70-75%) is extracted through a certain process and then to the pure product (content 90%), which is mainly used to synthesize coenzyme Q10(CoenzymeQl0) and vitamin K2. China is a big country of tobacco, the annual output of tobacco is more than 5000kt, so it is a huge material resource in China. With the continuous development of the tobacco industry, how to make better use of tobacco resources and improve its economic value is the subject and challenge we are facing. Tobacco leaves contain a variety of natural chemicals, many of which can be recycled, such as nicotine, lipids, essential oils, proteins, amino acids, cellulose, etc. Many of them are important biochemical and pharmaceutical raw materials. The comprehensive development and utilization of tobacco leaves began in foreign countries as early as the 2060s. It was only in the 2090s that China gradually began to extract and utilize solanesol from tobacco leaves. Therefore, extracting solanesol from tobacco (especially waste tobacco) can not only obtain an important pharmaceutical raw material, but also turn waste into treasure and increase the added value of the tobacco industry. |
| international market situation of high-purity solanesol | in recent years, the demand for high-purity solanesol in the international market has increased by 15% ~ 20%. the sales volume of solanesol in the international market was 4000 tons in 2004, 5200 tons in 2006 and 7500 tons in 2008. it is estimated that the demand for high-purity solanesol in the international market will reach 8000~10000 tons this year. Solanesol's hot sales benefited from its downstream product coenzyme Q10(CoQ10)'s rapid popularity in the international market. China is a large tobacco production country and has abundant raw materials for extracting solanesol. With the rapid improvement of solanesol extraction process year by year, several high-purity solanesol production lines have been under construction or trial production in Jilin, Jiangsu and other places recently. High-purity solanesol is the main intermediate for the synthesis of CoQ10, vitamin K2 and other best-selling raw materials. Western medical researchers have found that high-purity solanesol itself can be directly used as a clinical drug. The main clinical use is anti-heart failure, Treatment of liver damage and adjuvant treatment of cancer. According to the "Indian Medical Journal" report, many hospitals in India are currently conducting clinical controlled trials of "high-purity solanesol preparations as diabetes treatment agents and anti-cancer drugs. Once the above-mentioned new uses are recognized by the medical community, the total sales volume of high-purity solanesol in the international market will be greatly promoted in the future. In addition, CoQ10 synthesized by solanesol has also received great attention in the European and American medical circles, and its research results have been continuously reported, such as: it can promote oxidative phosphorylation and adenosine triphosphate (ATP) Synthesis, can be used as a cell metabolism and respiratory activator, help to enhance the function of the human immune system, etc. So far, CoQ10 has been included in the pharmacopoeia of the United States, Europe, Japan and other countries. In recent years, American medical researchers have also found that CoQ10 is compatible with statins for lowering blood lipids, which can greatly reduce the side effects such as rhabdomyolysis and prevent acute myocardial infarction in patients with coronary heart disease. It is worth mentioning that in today's frequent drug safety accidents, there have been no reports of CoQ10 toxic and side effects. In recent years, CoQ10 sales have grown rapidly in the United States, Australia, Germany, Italy, France and other European and American countries, as well as in Japan and my country. According to the latest statistics from abroad, the total sales volume of CoQ10 API in the world exceeded 1000 tons last year, covering drugs, cosmetics and health food. Therefore, CoQ10 is considered to be one of the "drugs with the greatest growth potential in the 21st century. It can be seen that whether it is used to synthesize CoQ10 or used as a drug, with the continuous deepening of research and practical application, high-purity solanesol will usher in the "golden age" of its market career ". |
| output analysis | according to statistics, the output of major CoQ10 API producers in the world in 2008 was: Japan, 600 tons; The United States, 200 tons; South Korea, 200 tons; Germany and Italy total 100 tons; China is about 30 tons. It is estimated that the actual global CoQ10 production last year was about 1200 tons. According to the calculation that 2 tons of high-purity solanesol raw materials are needed to produce 1 ton of CoQ10, the global high-purity solanesol used to synthesize CoQ10 alone reached about 2400 tons last year. Coupled with the consumption of high-purity solanesol for other purposes such as cosmetics and health food, it is estimated that the global total output of high-purity solanesol last year should be about 6000 tons. Since the crude solanesol extracted from tobacco leaves has the highest content of only 20%, it cannot be used as a raw material for the synthesis of CoQ10 or other drugs. Therefore, only by increasing its purity to 90% can it become a raw material for drug synthesis. At present, the price of high-purity solanesol in the international market is generally about US $300 per kilogram, and the average price of high-purity solanesol exported by my country is about US $250 per kilogram. However, in the past few years, China can only produce crude solanesol, and imports more than 1000 tons of high-purity solanesol raw materials from Japan every year for domestic synthesis of CoQ10 and other drugs. The most commonly used processes for producing CoQ10 are synthesis (the main raw material is high-purity solanesol) and microbial fermentation. Japan is one of the first countries in the world to master these two CoQ10 production processes, and it is also the main exporter of CoQ10 in the world. According to statistics, Japan's export of CoQ10 once accounted for about 90% of the total sales of CoQ10 in the international market. However, the main raw material for the synthesis of CoQ10, high-purity solanesol, was previously produced by Japan. With the enhancement of the ability of China and India to produce crude solanesol in large quantities, Japan has changed to import crude solanesol from China and India for the synthesis of CoQ10 and then exported to European and American markets after improving the purity. Data show that the annual export of solanesol to Japan accounted for about 60% of China's total crude solanesol exports. It is understood that although India's tobacco production is not as large as China's, its solanesol extraction industry was developed earlier than China's, and its solanesol products often compete fiercely with Chinese products in the international market. |
| use | is an important raw material for the synthesis of coenzyme Q10, which can be used as an activator of human cell respiration and metabolism and an immune enhancer for the body. Synthesis of coenzyme Q10 and vitamin K2 intermediates. C45 isopentenols are the most abundant lipids in tobacco leaves and are important precursors of tumorigenic polynuclear aromatic hydrocarbons in tobacco. anti-ulcer, anti-tumor, solanesol is an important pharmaceutical intermediate mainly used for the synthesis of coenzyme Q10 and vitamin K2 mainly used for the synthesis of coenzyme Q10 and vitamin K2. |
Last Update:2024-04-09 20:52:54
Supplier List
Multiple SpecificationsSpot supply
Product Name: Solanesol Visit Supplier Webpage Request for quotationCAS: 13190-97-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: solanesol Request for quotation
CAS: 13190-97-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 13190-97-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Solanesol Request for quotation
CAS: 13190-97-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13190-97-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Solanesol Visit Supplier Webpage Request for quotationCAS: 13190-97-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Solanesol Visit Supplier Webpage Request for quotationCAS: 13190-97-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: solanesol Request for quotation
CAS: 13190-97-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 13190-97-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Solanesol Request for quotation
CAS: 13190-97-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13190-97-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Solanesol Visit Supplier Webpage Request for quotationCAS: 13190-97-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



